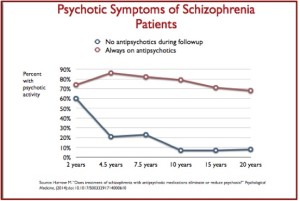
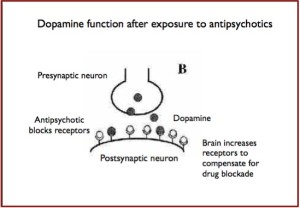
Broken Promises with Abilify

Based upon sales data for the world’s 15 top selling drugs, Abilify was ranked fourth, with $9.3 billion of sales in 2014. Reflect for a moment what this means; an antipsychotic drug had greater worldwide sales than Nexium (for acid reflux) and Crestor (for high cholesterol). While it is an approved medication in the US for bipolar 1 and schizophrenia, it is likely these sales figures reflect it use as an adjunct medication for major depression. Oh, and along with other antipsychotics, it is used off label to treat several other behavioral disorders such as Tourette’s and irritability associated with autism. This popularity is despite the reality that antipsychotics have a high incidence of negative side effects—greater than antidepressants and anti-anxiety medications.
The problems with antipsychotics have been known for a few years. As far back as 2010, Robert Field wrote: “Antipsychotic Medications Are Spelling Legal Trouble for Drugmakers” for the journal Pharmacy and Therapeutics. In 2007, Bristol-Myers Squibb paid $515 million to settle charges of illegally marketing Abilify for children and the elderly, “In contravention of FDA-approved labeling.” But that hasn’t been the end of legal troubles regarding Abilify. On May 3, 2016, the FDA published a safety announcement warning that Abilify (aripiprazole) can trigger impulse-control problems such as “compulsive or uncontrollable urges to gamble, binge eat, shop, and have sex.” These urges reportedly stop when the drug is discontinued or the dose reduced.
These compulsive behaviors can affect anyone who is taking the medicine. As a result, we are adding new warnings about all of these compulsive behaviors to the drug labels and the patient Medication Guides for all aripiprazole products.
The mechanism of action for Abilify is not clearly understood, but researchers think it over-stimulates dopamine 3 (D3) reward receptors, which are mainly located in the limbic system. This in turn triggers the compulsive behaviors. Gaboriau et al. reviewed case reports in Addictive Behaviors and indicated that pathological gambling (PG) appeared as quickly as a few days after Abilify was started; sometimes after an increase in dosage with 7 of the 8 case reports. PG then decreased after Abilify treatment was stopped or decreased; again sometimes as soon as a few days afterwards.
Limitations on the Gaboriau et al. findings included that most of the patients were already gambling before starting with Abilify. Several patients also had a history of substance use disorders. However, the authors noted that the same D3 receptor was implicated in another study by J. E. Ahiskog of the dopamine agonist medications, pramipexole and ropinirole, which are commonly used to treat Parkinson’s disease.
This hyperstimulation would apparently be particularly enhanced in cases of a previous treatment by antipsychotics acting as a dopaminergic receptors antagonist, owing to the up-regulation and the dopaminergic receptor hypersensitivity processes. The partial agonist action of aripiprazole then causes stronger effects. Moreover, the intrinsic dopamine pharmacodynamic activity of aripiprazole imparts it less action agonist than a complete agonist, which could explain why the occurrence of PG is sometimes late or due to dosage increase.
The above concern with Abilify was also supported by the findings of a study by Moore, Glenmullen and Mattison reported in JAMA Internal Medicine. Adverse drug event reports received by the FDA from 2003 to 2012 were reviewed for the six dopamine receptor agonist drugs marketed in the U.S. The review identified 1580 reports of impulse control disorder events, including pathological gambling, hypersexuality, compulsive shopping and others. They also detected weaker signals for antidepressants and antipsychotics.
The Daily Beast reported on a massive tort lawsuit being filed against Otuska and Bristol-Myers Squibb charging that Abilify created a compulsion for sex and gambling. Moreover, the suit claims the drug makers knew of the serious side effects because of required changes in Canadian and European warning labels, but waited for years to warn U.S. consumers. Thomas Moore of the Institute for Safe Medication Practices explained the drug triggers an urge to gamble constantly, sometimes with people with no prior interest. “It might be people starting to spend $300 a week on lottery tickets, and in other cases people will gamble away tens of thousands of dollars.” Moore went on to say:
We live in a society whose rules and laws assume people are responsible for their actions, including running up a large gambling debt. . . But we have scientific evidence that sometimes a drug can trigger a pathological urge to gamble so severe it can ruin someone’s life.
A woman who began using Abilify to aid in treating her PTSD developed a compulsive gambling problem. She used up her unemployment checks, pawned her husband’s automotive tools, and lied about needing money for baby formula. “Nothing was off-limits when it came to getting the money I needed to keep up the ruse.” She’d stuff her bed at night in order to fool her husband into thinking she was asleep when she was actually at the casino playing the slot machines.
Another woman developed hypersexuality. She started with online chatting with men. She became obsessed with sexual fantasies and took sexualized pictures of herself and sent them to select ‘friends.’ “I just couldn’t stop with the pictures and fantasies.” She also went on shopping sprees. Then her husband caught her. “The drug has destroyed my life, my reputation, and the lives of those I love.”
The website RxISK has multiple reports on adverse events with Abilify. “Abilify from the Inside Out” described bouts of akathisia (a state of agitation, distress, and restlessness), unusual aggression or anger, first time episodes of psychosis, suicidality, at least three confirmed suicides, movement disorders such as tremors, and (of course) compulsive gambling. The author said the reports were hard for him to read. Since most of the patients were on several meds, some patients couldn’t be sure that Abilify alone caused the problem. Even stopping Abilify was related to adverse drug events.
The above noted 2007 lawsuit, where Bristol-Myers Squibb paid $525 million to settle charges of illegal marketing, unveiled some of the marketing records for Abilify. Remember, one of the concerns was that it was illegally marketed for use with the elderly. The sales reps for Abilify would invite nursing home staff to picture a new resident, hunched in their chair, staring off into space because of ‘depression.’ “’Who wants to see that when they come to visit Mom on a Saturday?’ the reps would ask. ‘Wouldn’t we like to see her up and about, looking lively?’” The sale pitch worked. One woman wrote the following to RxISK:
I have seen many commercials about how drugs like Abilify can perk people right up. . . So I was not only disappointed and frightened by the results, but felt once again tricked and exploited by the big promises that drug companies make but never seem to keep.
I wish the above concerns weren’t true. But I’ve known individuals whose experiences on Abilify are consistent with the above discussion of its adverse effects. Sadly, even when sanctions are in the millions of dollars, the profits are higher. And it seems the cards are stacked against pharmaceutical companies being held accountable financially. So consumers have to fight against this by refusing to use Abilify and telling others what you have read here. If you are interested in other articles on the problems with Abilify and the other antipsychotics, try: “Antipsychotic Big Bang” or “Abilify in Denial” on this website.